Deadline
May 11, 2026
Funding Expanded! Increased prize pool and Stage 1 opportunities – creating more pathways for teams to advance.
Join an Upcoming Info Session →
$100M SMART Antiviral Prize to Advance Novel Antiviral Therapies
The SMART Antiviral Prize is focused on identifying safe and effective broad-spectrum small molecules that have the potential to advance into clinical trials and achieve U.S. regulatory approval. The goal of this competition is to advance the development of novel antivirals that strengthen the therapeutic pipeline and address gaps in strategic preparedness in partnership with innovators offering creative solutions.

Seeking Novel Therapeutic Solutions for Emerging Viral Diseases for Which No Treatments Exist
The SMART Antiviral Prize focuses on supporting the development of antivirals with broad-spectrum activity against members of the Togaviridae and/or Flaviviridae families.
There are currently no FDA-approved broad-spectrum antivirals for any viruses within these families—which include dengue, Zika, West Nile, and Chikungunya. These viruses collectively cause millions of infections each year, including increasing numbers of cases in the United States.
CONCEPT
Concept Stage
$20M
Up to 8 prizes, $2.5M each
CLOSED
No longer accepting applications
CLOSED May 11
STAGE 1
Hit-to-Lead
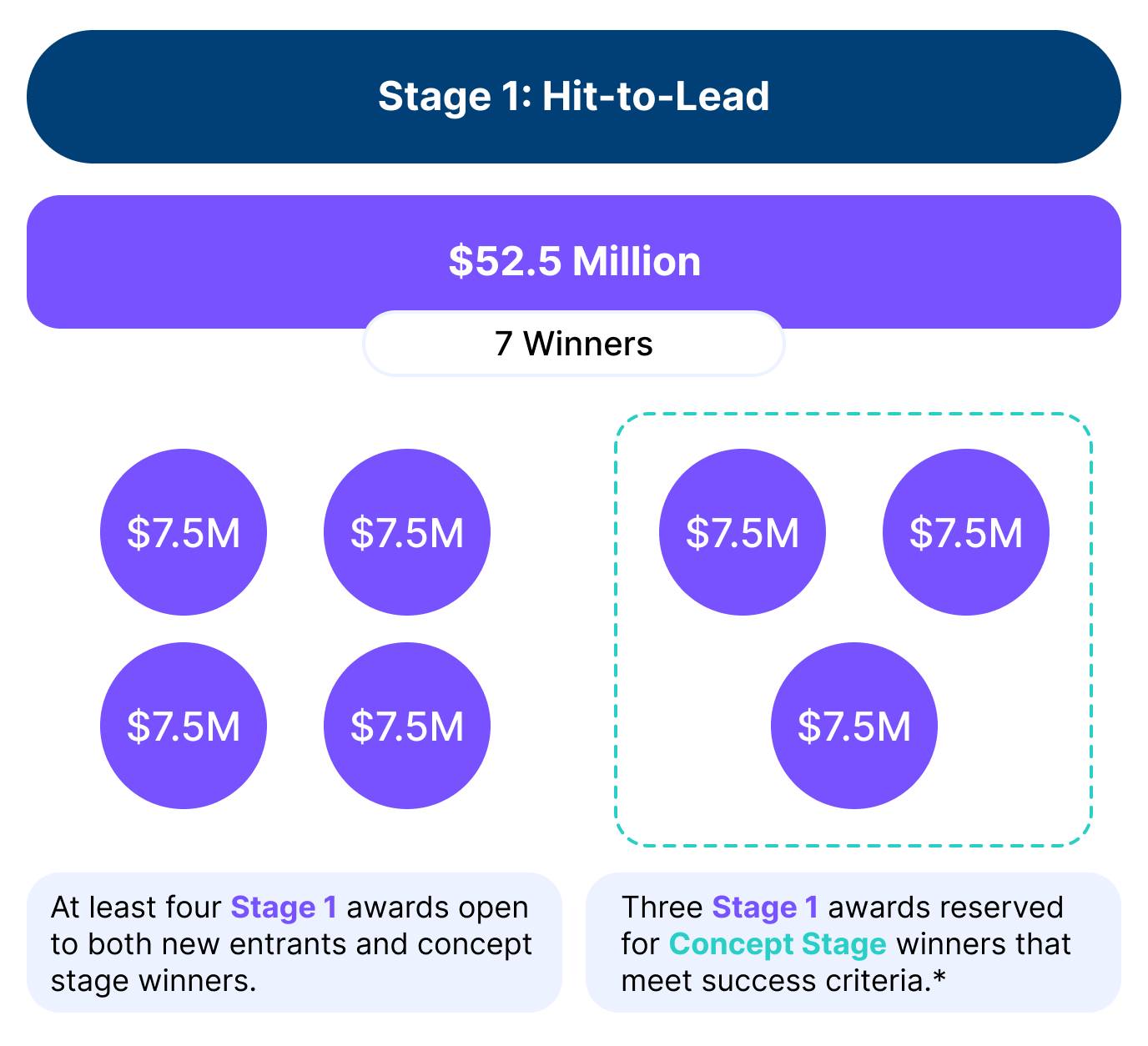
$52.5M
Up to 7 prizes, $7.5M each
Now with Increased Funding!
Opens Summer 2026
Summer 2026
STAGE 2
Lead Optimization
TBD
At least 4 prizes
Opens Early 2028
Early 2028
STAGE 3
IND Readiness
TBD
At least 2 prizes
Opens Early 2030
Early 2030
Concept Stage
The Concept Stage application period for the SMART Antiviral Prize is now closed.
Our review panel is currently evaluating submissions from innovators developing next-generation broad-spectrum antiviral approaches. Stage 1: Hit-to-Lead is expected to open this summer and will support teams advancing promising antiviral candidates through early optimization and validation milestones.
Additional details, eligibility information, and timelines will be announced soon.
Desired Product Attributes
This document outlines the Desired Product Attributes that applicants may find useful when shaping antiviral concepts for submission to the SMART Antiviral Prize.
Evaluation Process & Criteria
The SMART Antiviral Prize uses a two-stage review process. This document outlines the Evaluation Process & Evaluation Criteria for the Concept Stage.
FAQs & Resources
Review frequently asked questions, program stage overviews, and general resources for the SMART Antiviral program Concept Stage engagement and application.
Submit Application
Apply now to submit your Concept Stage antiviral innovation tor funding and support through the SMART Antiviral Prize.
Turning Breakthrough Science Into Life-Saving Antiviral Therapies
The SMART Antiviral Prize is more than a competition – it’s a call to advance bold ideas and move promising science forward.
Rewarding Translational Scientific Breakthroughs Using a Tiered, Sequenced Prize Design
Participation in the SMART Antiviral Prize is subject to official Terms & Conditions.
- The Goal
The SMART Antiviral Prize is focused on identifying safe and effective broad-spectrum small molecules that have the potential to advance into clinical trials and achieve U.S. regulatory approval. The goal of this competition is to advance the development of novel antivirals that strengthen the therapeutic pipeline and address gaps in strategic preparedness in partnership with innovators offering creative solutions.
- A Multi-Stage Competition
The SMART Antiviral Prize is a multi-stage competition designed to move candidates from idea to IND readiness through clear, stage-specific milestones and published evaluation criteria. It begins with a Concept Stage that prioritizes a well-defined technical and development approach (not extensive data), with later stages requiring progressively deeper technical submissions and evidence of broad-spectrum activity and product attributes—while allowing multiple entry points as candidates advance.

Teaming Opportunity
If you or your organization would like to explore collaborations with other innovators for potential joint collaborative applications, please visit the SMART Antiviral Prize Teaming Page.
Upcoming Info Sessions & Webinars
Have questions about the SMART Antiviral Prize? Join one of our upcoming info sessions to learn more about the challenge, the application process, and what we’re looking for. You’ll also have the opportunity to engage directly with the VITAL team during a live Q&A.

Call for Antiviral Testing Partners
Do you have capabilities in running live-virus, cell-based antiviral potency assays for Dengue and Chikungunya and are interested in serving as a reference facility for the SMART Antiviral Prize?
Disclaimer
All information provided within is preliminary and subject to change. Specific criteria and requirements are currently being refined. In the event of any inconsistency, the official SMART Antiviral Prize Terms & Conditions supersede this information.
Objectives for Each Stage of the SMART Antiviral Prize
Concept Stage | CLOSED
Describe development plans to discover or advance broad-spectrum small molecule antivirals for Flaviviridae and/or Togaviridae families.
Stage 1: Hit-to-Lead
Identify at least one promising chemical series with reproducible cell-based antiviral activity and an early SAR that supports rational optimization.
Build an initial “go/no-go” dataset for each series—basic ADME/PK, key developability flags, and early safety/DDI triage—to justify moving into lead optimization.
Stage 2: Lead Optimization
Optimize potency, selectivity, and exposure to produce a well-characterized lead and backup, with a clear plan to mitigate remaining liabilities.
Demonstrate in vivo proof-of-concept efficacy in a relevant animal model and nominate an IND candidate (and backup where feasible) suitable for IND-enabling studies.
Stage 3: IND Readiness
Complete the nonclinical package required for first-in-human dosing, including GLP toxicology/safety pharmacology and definitive ADME/DDI studies, plus manufacturing/CMC readiness (quality, stability, and formulation appropriate for clinical use).
Conduct appropriate FDA interactions and assemble an IND-ready submission package suitable for filing to initiate clinical trials.
A Multi-Stage Model to Accelerate Antiviral Innovation
The SMART Antiviral Prize offers up to $100M in awards to support innovators across four progressive stages – from early concept through IND readiness. Each stage builds upon prior success, rewarding the most promising candidates for achieving ambitious yet attainable milestones on an accelerated timeline.
Concept Stage

- Total Prize Pool
$20M
- Number of Prizes
Up to eight – $2.5M each
- Stage Launch & Declaration of Intent
Closed May 11, 2026
Stage 1: Hit-to-Lead

- Total Prize Pool
Up to $52.5M
- Number of Prizes
Up to seven – up to $7.5M each
- Stage Launch & Declaration of Intent
Anticipated Summer 2026
Stage 2: Lead Optimization

- Total Prize Pool
TBD
- Number of Prizes
At least four
- Stage Launch & Declaration of Intent
Anticipated Early 2028
Stage 3: IND Readiness

- Total Prize Pool
TBD
- Number of Prizes
At least two
- Stage Launch & Declaration of Intent
Anticipated Early 2030
Award Allocation across Concept Stage and Stage 1: Hit-to-Lead
The SMART Antiviral Prize fosters the growth of early-stage innovators by providing seed funding at the Concept Stage, and by reserving two awards in Stage 1 for Concept Stage winners. Teams with candidates already in preclinical development can enter Stage 1 without having participated in the Concept Stage.

Awards for Concept Stage
Up to eight awards will be made after a merit-based review of all proposals. Submissions will be reviewed as a batch after the solicitation period is closed. Applications will be accepted starting in early February 2026. Concept Stage award decisions are anticipated in Q3 2026. The evaluation process and accompanying criteria are outlined below.
Awards for Stage 1: Hit-to-Lead
Up to seven awards will be made in total. Up to three (3) of the seven awards are reserved exclusively for Concept Stage winners that meet the Optimal or Essential Success Criteria within the specified submission window.* All applicants must submit a Declaration of Intent and receive approval before their submissions can be reviewed.
Rolling Review (First-to-Finish)*: Data packages may be submitted at any time during the submission window (anticipated to open in early 2027). Submissions will be reviewed in the order received, and awards will be made on a first-to-finish basis, subject to validation of the data.
Two-Success Criteria Tracks: Data packages may be submitted against one of two success criteria: Optimal Success Criteria or Essential Success Criteria, as defined in the Target Compound Profile.
- Optimal Track: Awards for Optimal submissions will be made on a rolling basis as soon as the data are reviewed and validated, until all available prizes for Stage 1 (that have not been reserved for Concept Stage winners) are awarded.
- Essential Track: Submissions that meet only the Essential Success Criteria will be held and considered at the end of the submission period. Essential awards will be made only if funding remains after all Optimal awards have been issued.
Reserved Awards for Concept Stage Winners: The three reserved awards are available only to eligible Concept Stage winners and may be earned at any time during the submission window. These reserved awards will be issued to the first three Concept Stage winners whose data packages meet the Optimal Success Criteria. If none of the Concept Stage winners meet the Optimal Success Criteria, the reserved awards will be issued to the first three Concept Stage winners to meet the Essential Success Criteria during the submission window.
Awards for Concept Stage
Up to eight awards will be made after a merit-based review of all proposals. Submissions will be reviewed as a batch after the solicitation period is closed. Applications will be accepted starting in early February 2026. Concept Stage award decisions are anticipated in Q3 2026. The evaluation process and accompanying criteria are outlined below.
Awards for Stage 1: Hit-to-Lead
Up to six awards will be made in total. Up to two (2) of the six awards are reserved exclusively for Concept Stage winners that meet the Optimal or Essential Success Criteria within the specified submission window.*All applicants must submit a Declaration of Intent and receive approval before their submissions can be reviewed.
Rolling review “first-to-finish”
Data packages may be submitted at any time during the submission window (anticipated to open in early 2027). Submissions will be reviewed in the order received, and awards will be made on a first-to-finish basis, subject to validation of the data.
“Reserved awards” for Concept Stage winners
The two reserved awards are available only to eligible Concept Stage winners and may be earned at any time during the submission window. These reserved awards will be issued to the first two Concept Stage winners whose data packages meet the Optimal Success Criteria. If none of the Concept Stage winners meet the Optimal Success Criteria, the reserved awards will be issued to the first two Concept Stage winners to meet the Essential Success Criteria during the submission window.
Two success- criteria tracks
Data packages may be submitted against one of two success criteria: Optimal Success Criteria or Essential Success Criteria, as defined in the Target Compound Profile.
Optimal track:
Awards for Optimal submissions will be made on a rolling basis as soon as the data are reviewed and validated, until all available prizes for Stage 1 (that have not been reserved for Concept Stage winners) are awarded.
Essential track:
Submissions that meet only the Essential Success Criteria will be held and considered at the end of the submission period. Essential awards will be made only if funding remains after all Optimal awards have been issued.
Rolling review “first-to-finish”
Data packages may be submitted at any time during the submission window (anticipated to open in early 2027). Submissions will be reviewed in the order received, and awards will be made on a first-to-finish basis, subject to validation of the data.
Two success- criteria tracks
Data packages may be submitted against one of two success criteria: Optimal Success Criteria or Essential Success Criteria, as defined in the Target Compound Profile.
Optimal track:
Awards for Optimal submissions will be made on a rolling basis as soon as the data are reviewed and validated, until all available prizes for Stage 1 (that have not been reserved for Concept Stage winners) are awarded.
Essential track:
Submissions that meet only the Essential Success Criteria will be held and considered at the end of the submission period. Essential awards will be made only if funding remains after all Optimal awards have been issued.
“Reserved awards” for Concept Stage winners
The two reserved awards are available only to eligible Concept Stage winners and may be earned at any time during the submission window. These reserved awards will be issued to the first two Concept Stage winners whose data packages meet the Optimal Success Criteria. If none of the Concept Stage winners meet the Optimal Success Criteria, the reserved awards will be issued to the first two Concept Stage winners to meet the Essential Success Criteria during the submission window.
Concept Stage Evaluation Process & Criteria
Overview
The SMART Antiviral Prize uses a two-stage review process:
- Written Submission Review
- Written Submission Review and Virtual Presentation (Pitch Call)
Stage 1: Written Submission Review
An expert judging panel will review, score, and rank all Concept Stage written submissions using the published evaluation criteria. Scores and rankings will be based solely on the information provided in the written submission at the time of submission.
Stage 2: Virtual Presentations (“Pitch Calls”)
Based on rankings from Stage 1, the highest-scoring applicants will be invited to participate in a virtual presentation (“Pitch Call”) with the judging panel. The Administrator expects to invite fifteen (15) applicants. To support a balanced portfolio, the Prize Administrator intends to invite at least two (2) applicants targeting each prioritized viral family (Togaviridae and Flaviviridae).
Prize Award Recommendations
Following Pitch Calls, judges will submit results—reflecting final scoring and prize recommendations—to Start2 for decision. Consistent with the stated evaluation criteria and eligibility requirements, the final recommendations will support selection of eight (8) winners with at least one (1) winner from each prioritized viral family (Togaviridae and Flaviviridae).
Evaluation Criteria
The submissions will be reviewed using the following evaluation criteria.
Scientific rationale and antiviral target strategy
Judges will assess how clearly the submission explains its antiviral concept, including the proposed mechanism of action, relevance to broad-spectrum activity within one or more prioritized viral families, and strength of the supporting evidence (e.g., literature, public data, or preliminary in silico/experimental findings) that the target or pathway is essential, druggable, resistant to development of viral escape, and plausibly conserved across multiple viruses.
Development and regulatory strategy
Judges will consider how well the submission lays out a realistic, coherent path from the current state of the program toward a Phase 1–ready small-molecule antiviral candidate, including key preclinical activities and milestones, anticipated regulatory needs, and a thoughtful approach to identifying and mitigating major technical, regulatory, and operational risks over the anticipated prize stages.
Capabilities, partnerships, and execution feasibility
Judges will evaluate whether the entrant and any proposed partners have the expertise, resources, and collaborations needed to execute the plan, including relevant scientific and development capabilities, access to required infrastructure and IP/freedom to operate, and an overall program management approach that makes timely, high-quality delivery of the proposed work feasible.
In Stage 2 of the evaluation process, in addition to the written submission, judges will consider the following factors during the Pitch Call:
- Consistency with the written submission
- Clarity regarding key risks and proposed mitigation strategies
- Feasibility of near-term milestones
- Responsiveness to judges’ questions

Target Compound Profile Measuring Success in Stage 1: Hit-to-Lead
The SMART Antiviral Prize has outlined the set of target criteria for eligible entrants to reach by the end of Stage 1. Applicants applying for Concept Stage funding should design a credible development plan with these milestones in mind to address early risks and reach the project goals within the data submission period.
Disclaimer
All information provided within is preliminary and subject to change. Specific criteria and requirements are currently being refined. In the event of any inconsistency, the official SMART Antiviral Prize Terms & Conditions supersede this information.
Desired Product Attributes for Small Molecule Broad-spectrum Antivirals
This document outlines Desired Product Attributes that applicants may find useful when shaping antiviral concepts for submission to the SMART Antiviral Prize. These attributes are provided as a draft and are subject to change as the prize design is further refined.
Attribute
Minimal
Ideal
Indication for Use
- Treatment of acute, laboratory-confirmed infection caused by viruses within the Flaviviridae and/or Togaviridae family
- Treatment and prophylaxis
- Family wide label supported by bridging across a prespecified breadth panel
Mechanism of Action
- Direct-acting antiviral (targets highly conserved viral factors) or indirect acting antiviral (targets host factors required for viral entry, replication, and/or persistence)
Antiviral Activity (Breadth and Resistance)
- EC50 ≤1 µM across all viruses in a predefined breadth panel
- Documented, acceptable level of resistance development with understanding of potential resistance across viral species
- EC50 ≤100 nM across all viruses in a predefined breadth panel; EC50 ≤1 µM across ALL pathogenic viruses within the family
- No evidence of emergence of resistance in clinical trials
Target Population
- All populations, e.g., pediatrics (incl. neonates), adults, older adults, pregnant/lactating, immunocompromised
Contraindications, Use, Adverse Reactions and Drug Interactions
- Acceptable adverse event rate vs. benefit; manageable toxicity
- No black box warning or major precautions
- No embryo-fetal toxicity
- Some contraindications tolerated
- No severe interactions with routine drugs; monitor CYP pathway; may need dose adjustment with other medications
- Compatible with potential standard of care (SOC) options
- Well tolerated, mild or no side effects
- No significant contraindications or DDIs; compatible with most therapies
Clinical Efficacy
- Clinically meaningful improvement in time to symptom resolution for acute infections when compared to untreated patients
- Clinically meaningful reduction in symptom severity/progression to severe disease or hospitalization and strong prevention of progression or transmission, if applicable
- Prevention of chronic symptoms/post-viral sequelae
- Safety and tolerability profile supports evaluation for prophylactic indications
Clinical Pharmacology
- Defined PK/PD; dosing maintains EC90 in target tissue(s)
- Antiviral activity (e.g., protein- adjusted 90% effective concentration pa-EC90)) supporting a PK/PD Index (PDI) ≥3 (e.g., predicted Ctrough/pa- EC90)
- Predictable and consistent PK/PD in all sub-populations; long half-life
- Dosing maintains 3x EC90 in target tissue(s)
- Antiviral activity supporting PDI≥10 where feasible
Therapeutic Modality
- Small molecule (at or below 900 daltons)
Therapeutic Window
- Within 48 hours from symptom onset
- Maintains effectiveness when started 5 days from symptom onset; effective even with delayed diagnosis
- Suitable for treatment with extended protection providing sustained viral clearance for persistent infections
Dose Regimen
- 3 doses/day for up to 14 days, or as needed based on diseasei
- Single dose
Route of Administration
- Oral
- Multiple routes, including oral and parenteral
Storage and Stability
- ≥ 2 years at 2-8˚C and/or RT, stable in field conditions
- ≥ 3 years at controlled RT, compatible with pandemic deployment
Regulatory Path, Registration
- NDA
iPersistent infections caused by Chikungunya lead to prolonged viral presence (up to 28 days) and may need longer duration of treatment for sustained viral clearance.

Target Compound Profile Measuring Success in Stage 1: Hit-to-Lead
The SMART Antiviral Prize has outlined the set of target criteria for eligible entrants to reach by the end of Stage 1. Applicants applying for Concept Stage funding should design a credible development plan with these milestones in mind to address early risks and reach the project goals within the data submission period.
Disclaimer
All information provided within is preliminary and subject to change. Specific criteria and requirements are currently being refined. In the event of any inconsistency, the official SMART Antiviral Prize Terms & Conditions supersede this information.

SMART Antiviral Prize Teaming
Thank you for your interest in the SMART Antiviral Prize. Collaborations between diverse innovators are encouraged to help accelerate the development of broad-spectrum, small molecule antiviral therapies.
Submissions for the Concept Stage are now closed, but participant profiles remain available for those looking to connect with others in the field. Sort by profile activity or filter by area of expertise to find relevant collaborators.
Check back regularly for updates and new opportunities.
Last updated:
Synko Pharma Corp. seeks to develop broad spectrum antivirals based on their synthetic carbohydrate receptor (SCR) technology. SCRs are small molecules with molecular weights below 900 that operate by inhibiting the role of envelope glycans in the viral lifecycle, and we have already demonstrated in vitro and in vivo broad spectrum antiviral activity, including against multiple flaviviridae.
Synko Pharma Corp. focuses on developing broad spectrum antivirals, and the current stage of development is TRL3/4 based on the medical countermeasures roadmap. This includes identifying and validating a novel target (viral envelope glycans), in vitro activity against live viruses, and assays to demonstrate that the origin of antiviral activity of our countermeasures is glycan binding.
Synko will be seeking partners for clinical formulations, non-GMP and GMP manufacturing, and running clinical trials.
IIT Research Institute (IITRI)
Last updated:
Contact
Location
Chicago, Illinois
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
IITRI is a midsized nonprofit preclinical CRO with extensive experience and a long history of supporting antiviral development. We are a fully accredited facility with ABSL-2 and ABSL-3 containment suites available for studies involving pathogenic agents and medical countermeasures, and inhalation exposure suites available for studies in small and large animals. Our GLP-compliant bioanalytical laboratory is onsite for efficient and transport-free coordination of study teams and samples.
Current Research Focus Areas
IITRI specializes in infectious disease vaccine and therapeutic development, IND-enabling programs, and inhalation toxicology for biotech and government sponsors. We have experience working with members of the Togaviridae and Flaviviridae families, along with many other emerging and relevant pathogens. Active research areas include aerosol transmissible pathogens, therapeutic response characterization, and evaluation of antiviral delivery platforms across our multidisciplinary scientific teams.
What We’re Seeking in Teaming Partners
IITRI is interested in collaborating with organizations developing innovative antiviral platforms or strategies that require infectious challenge models and comprehensive safety programs. We can support exploratory, non GLP infectious disease evaluations in ABSL 2 and ABSL 3 environments, and fully GLP compliant toxicology and pharmacology studies. Our integrated scientific teams provide continuity across discovery, proof of concept, and data for IND submissions. We seek partners who value direct scientific engagement and a collaborative approach to advancing antiviral and biodefense technologies.
NanoViricides, Inc.
Last updated:
Contact
Location
Shelton, CT
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- CMC / Formulation Manufacturing
Relevant Technology
NanoViricides is developing antiviral drugs using a novel nanomedicines technology. For SMART Prize Project#1, we intend to design & develop novel pan-Flavivirus Entry-Inhibitors that would disturb virus surface. In Project#2, we plan to design & develop novel pan-RNA virus RDRP-suicide-inhibitors. We have strong in-house capabilities for chemistry, synthesis, scale-up, cGMP manufacture, & clinical-scale product fill-and-finish. We depend upon collaborators for Non-clinical studies.
Current Research Focus Areas
Lead direct-acting, cell-mimetic, broad-spectrum antiviral, NV-387: Strong in animal models vs disparate viruses; Influenza, RSV, CoronaV, OrthopoxV, Measles. Ph I successful. Ph II vs MPox scheduled, DRC. TRL4/5.
NV-HHV-1, HVEM-mimetic pan-herpesvirus drug. NV-HIV-1, pan-HIV CD4 & CCR5 sites presenting, presumably functional cure. TRL3/4.
Small molecules – Various broad-spectrum RDRP-inhibitory nucleotide analogs; NV-485, TRL3, successful animal models RSV, Measles; expect TogaV& FlaviV also.
NV-HHV-1, HVEM-mimetic pan-herpesvirus drug. NV-HIV-1, pan-HIV CD4 & CCR5 sites presenting, presumably functional cure. TRL3/4.
Small molecules – Various broad-spectrum RDRP-inhibitory nucleotide analogs; NV-485, TRL3, successful animal models RSV, Measles; expect TogaV& FlaviV also.
What We’re Seeking in Teaming Partners
In vitro (including BSL3) and In vivo antiviral assays & models for efficacy and infected species safety tolerability studies. Viruses of interest: Flaviviridae, mainly DENV-1,2,3,4, ZikaV, YFV, WNV incl. Clinical isolates, for Project#1 Entry Inhibitors. For Project#2 RDRP Inhibitors, Togaviridae: CHIKV, RossRV, BarmahFV, also BSL3 Enceppalitic viruses. DMPK studies including DDI, PPB, SMIL, Metabolic. Non-GLP and GLP PK studies. IND-enabling Non-clinical Safety-Tox and other studies.
Brown Lab: University of Florida
Last updated:
Contact
Location
Orlando, FL
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
We help de-risk and accelerate antiviral development using advanced PK/PD-driven infection models and translational modeling. Our team designs dosing and combination strategies that maximize efficacy, limit resistance, and minimize toxicity for medically important viruses, including flaviviruses and togaviruses, with full BSL-2/ABSL2 and BSL-3/ABSL3 select-agent capabilities.
Current Research Focus Areas
We are able to use our PK/PD infection and mathematical models to design optimal dosing strategies for antiviral agents that will improve therapeutic outcomes in patients. We are able to de-risk development by identifying the optimal dose and dosing interval for agents as monotherapy or in combination that will maximize efficacy and prevent resistance emergence with minimal toxicity. We use in vitro and small rodent in vivo models. Our labs are certified for BSL-2 up to select agent BSL-3 work
What We’re Seeking in Teaming Partners
We seek teaming partners with identified antiviral hit or lead compounds and complementary discovery or development expertise. Our ideal collaborators contribute medicinal chemistry and clinical translation capabilities to support rapid PK/PD optimization and advancement toward first-in-human studies.
GiwoTech Inc.
Last updated:
Contact
Location
Boston, MA
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
GiwoTech is building an AI-driven platform that simulates protein dynamics and molecular interactions at atomic resolution to accelerate precision drug discovery and transform previously undruggable targets into viable therapeutics.
Current Research Focus Areas
An AI-driven molecular simulations platform to create digital twins of biological systems, focusing on protein-protein interactions and viral mechanisms. Our physics-informed machine learning simulations decodes protein dynamics at atomic resolution to unlock previously undruggable targets for therapeutic discovery. Currently NSF STTR Phase I-funded with BARDA support, advancing core simulation engine development to compress drug discovery timelines from years to months.
What We’re Seeking in Teaming Partners
We seek collaborators with expertise in: (1) experimental viral characterization and BSL-3 assay development for validation of computational predictions; (2) pharmaceutical manufacturing and GMP-scale production of antiviral therapeutics; (3) clinical trial design and regulatory expertise for FDA pathway navigation. Partners should bring existing pharma relationships, translational biology validation pipelines, o bridge computational predictions to clinical outcomes.
Allegheny Biosciences, LLC
Last updated:
Contact
Location
Lexington, KY
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
Computational drug discovery services spanning scaffold-based generative AI, goal-directed molecular optimization, and ADMET prediction. Multiple generative architectures and tools are available depending on project needs, with models fine-tuned for specific viral targets as required. The approach combines AI-driven exploration with medicinal chemistry knowledge to generate practical molecules against targets of interest.
Current Research Focus Areas
Antiviral target analysis and small molecule design for Flavivirus and Alphavirus families. Broad domain expertise spanning clinical pharmacology, structural biology, toxicology, and drug metabolism informs the computational work. Current infrastructure supports de novo generation, hit expansion, multi-objective optimization, and ADMET property prediction.
What We’re Seeking in Teaming Partners
Collaborators with virology expertise and antiviral assay capabilities to validate computational predictions. Ideal partners have experience testing compounds against Togaviridae or Flaviviridae targets. Synthetic chemistry access is helpful but can be addressed through CROs as needed. Computational hit generation, optimization, and property prediction offered in return. Open to joining established teams or forming new collaborations.
Atea Pharmaceuticals
Last updated:
Contact
Location
Boston, MA
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
Relevant Technology
Atea is a clinical stage biopharmaceutical company working to address unmet medical needs with innovative oral antiviral therapies targeting viral RNA-dependent RNA polymerases. With a platform of more than 2500 nucleoside and nucleotide prodrugs with extensive SAR, we can identify highly potent antivirals such as AT-2490 – a broad-spectrum polymerase inhibitor against both flaviviruses and togaviruses. Ongoing synthesis of new compounds through SAR may lead to new potential candidates.
Current Research Focus Areas
Broad spectrum nucleotide with demonstrated nanomolar in vitro activity against Dengue, Zika, Yellow Fever, Japanese Encephalitis, West Nile, Powassan, and Chikungunya. A potential clinical candidate, AT-2490, has been identified to progress into IND enabling studies to include additional testing against Flavivirus and Togavirus genotypes/serotypes/strains. Test in relevant animal efficacy models and other IND enabling activities including DMPK, nonclinical toxicology, and CMC development.
What We’re Seeking in Teaming Partners
In vitro (including BSL-3 labs) and in vivo testing (in relevant animal models, such as non-human primates for Chikungunya) against viruses to include Dengue, Zika, Yellow Fever, West Nile, Powassan, and Chikungunya. DMPK studies including in vitro DDI, PPB, metabolic stability, and cross-species metabolic ID in hepatocytes. Non-clinical toxicology studies in 2 species, GLP safety pharmacology and toxicology.
Southern Research: A Non-profit Research Institute & Full-Service CRO
Last updated:
Contact
Location
Birmingham, AL
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
Mission-driven nonprofit research institute: Southern Research is an 85-year-old nonprofit with ~200 employees translating scientific discovery into real-world impact through partnerships with government, industry, and academia. Academic and clinical integration: Southern Research leverages direct access to UAB’s core laboratories, clinical trial networks, and clinician-scientist expertise - reducing development risk and accelerating translation from discovery into and through clinical trials.
Current Research Focus Areas
Full service nonclinical CRO with deep expertise and experience in alphaviruses and flaviviruses. Federal select agent program; A/BSL-3 facilities and laboratories. AAALAC accredited with small and large animal capacity, including NHPs. End-to-end small molecule discovery from target identification to clinical lead. Advanced and complex synthetic chemistry capabilities. High-throughput screening and automation/robotics infrastructure. Early and late-stage clinical trial sample testing (GCLP).
What We’re Seeking in Teaming Partners
Novel, high-impact antiviral targets suitable for small-molecule discovery and optimization programs. Chemical assets or scaffolds with potential for maturation into differentiated clinical leads and backup candidates. Phenotypic discovery opportunities enabling development of novel antiviral or host-directed therapeutic programs. Antiviral or host-directed programs requiring integrated nonclinical support, including non-GLP and GLP studies. A/BSL-4 capabilities to complement our high-containment virology and translational research capabilities.
Artemis Bioservices
Last updated:
Contact
Location
Delft, The Netherlands
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
Artemis Bioservices is a GLP‑certified CRO supporting BARDA‑aligned development of direct‑acting antivirals with deep expertise in Flaviviridae and Togaviridae. Artemis provides in‑vitro antiviral efficacy testing, viral load reduction, and live and pseudotyped virus neutralization assays to assess family‑wide breadth, mechanism‑of‑action and resistance risk. Artemis has supported and executed multiple IND‑ready antiviral programs from discovery through clinical stages.
Current Research Focus Areas
Our primary focus is providing GLP‑certified virology and immunology services as a subcontracting CRO supporting BARDA‑aligned antiviral programs. Artemis Bioservices specializes in translational in‑vitro and ex‑vivo antiviral efficacy testing, with deep expertise in Flaviviridae and Togaviridae. We support partners from early discovery through IND‑ready stages by delivering robust data on antiviral activity, mechanism‑of‑action, resistance risk and correlates of protection.
What We’re Seeking in Teaming Partners
We seek teaming partners developing direct‑acting antivirals who require robust, BARDA‑aligned preclinical datasets. Ideal partners bring antiviral assets, chemistry, PK/PD, or clinical development expertise, while Artemis provides GLP virology and immunology services covering efficacy, breadth, and resistance risk. We have supported multiple startup companies in building high‑quality datasets that enabled successful investor engagement and competitive funding and partnering outcomes.
Battelle
Last updated:
Contact
Location
Columbus, Ohio
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- AI / ML
- Virology & Infection Models
Relevant Technology
Battelle provides capabilities to support antiviral development against viruses from the Flaviviridae and Togaviridae families, including existing in vitro and in vivo models for efficacy assessments and expertise in the development of such models to assess novel technologies. Our BSL-3+ containment facility is the largest privately maintained in the US and is operated under a well-established quality management system, and we provide a team of experts to support rapid evaluation of MCMs.
Current Research Focus Areas
Battelle has capabilities to support throughout product development using an established roadmap demonstrated by successful support of multiple product licensures via FDA Animal Rule. Our specialized capabilities being preclinical development supporting in vitro and in vivo efficacy assessments.
What We’re Seeking in Teaming Partners
Battelle seeks potential teaming partners in need of virology expertise and laboratory infrastructure (up to enhanced BSL-3) to support small molecule development and evaluation by being a partner with high quality, proven track record supporting therapeutic development against emerging infectious diseases spanning TRL-1 to TRL-7/8.
MRC University of Glasgow Centre for Virus Research
Last updated:
Contact
Location
Glasgow, Scotland
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- AI / ML
- Virology & Infection Models
Relevant Technology
The MRC Centre for Virus Research is a leading UK institute studying human and animal viruses. Its research spans virus biology and evolution, sequencing, bioinformatics, and viral genomics. It is a standing national capability for virus discovery, preparedness and response, enabling rapid progression from early detection to actionable innovations to tackle viral threats in the UK and worldwide.
Current Research Focus Areas
Within the MRC Centre for Virus Research Translational Hub, high-throughput screening assays have been established to enable identification of novel antiviral candidates. Supported by high-containment laboratories, automated robotics, and advanced in-vitro and in-vivo infection models, the Hub enables pre-clinical evaluation of therapeutics, diagnostics, and vaccines, strengthening preparedness and global response to viral diseases, including Flaviviridae and Togaviridae.
What We’re Seeking in Teaming Partners
To complement the virology expertise at the MRC Centre for Virus Research, the Centre is seeking partnerships with medicinal chemists who can provide compound libraries or candidate molecules for in-vitro screening, compounds suitable for in-vivo efficacy studies, and advanced drug assets ready for comprehensive in-vivo validation, SAR studies and translational progression. Partners who can provide GLP/GMP capabilities.
Coley Lab: Massachusetts Institute of Technology
Last updated:
Contact
Location
Cambridge, Massachusetts
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- AI / ML
Relevant Technology
Various generative molecular design methods with broad relevance to small molecule drug discovery. In particular, methods for synthesizable molecular design (https://www.arxiv.org/abs/2512.00384) and interaction-aware 3D molecular design (https://arxiv.org/abs/2411.04130).
Current Research Focus Areas
Many AI/ML workflows for molecular design validated on computational tasks or retrospective benchmarks, with a handful of prospective experimental projects.
What We’re Seeking in Teaming Partners
Interested in partnering to bring our AI approaches to hit expansion and molecular optimization to teams that have the other pieces of the puzzle. We have no virology expertise or assay capabilities and are not in a position to lead a team.
Ai-biopharma
Last updated:
Contact
Location
Montpellier, Occitanie, France
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
Ai-biopharma seeks to develop broad spectrum direct acting antivirals based on their chemical libraries (physical and virtual) of viral polymerase inhibitors (active site) bearing broad viral coverage, as well as its medicinal chemistry expertise of polymerase inhibitors and proprietary chemoinformatic platform (AI/ML).
Current Research Focus Areas
Lean biotech at drug discovery stage (H2L) developing its own projects of viral polymerase inhibitors. The company is expanding virus coverage of its RNA virus inhibitors' library. Performing chemoinfo, drug design, medicinal chemistry optimization, chemical syntheses, and physchem props assessment in house.
What We’re Seeking in Teaming Partners
Ai-biopharma is looking for 3 main resources (collaborative or FFS/CRO):
1-Antiviral classical cell screening for both Flaviviridae and Togaviridae viruses of interest as well as primary cell assays on some hits and POC study in infected animal model.
2-ADMET and derisking testing to move to stage 1.
3-Project coordination if collaborative project (non-FFS/CRO).
1-Antiviral classical cell screening for both Flaviviridae and Togaviridae viruses of interest as well as primary cell assays on some hits and POC study in infected animal model.
2-ADMET and derisking testing to move to stage 1.
3-Project coordination if collaborative project (non-FFS/CRO).
Nanyang Technological University
Last updated:
Contact
Location
Singapore, Singapore
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
Relevant Technology
Molecular basis of Flavivirus and Togavirus replication mechanisms
Current Research Focus Areas
Understanding of the molecular mechanism of virus replication process and its inhibition by small molecule compounds
What We’re Seeking in Teaming Partners
Biochemistry, enzymology, structural biology, and virology
Collaborative Community Against Coronavirus CIC
Last updated:
Contact
Location
London, England
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Development / ADME / Safety
Relevant Technology
3C is a UK-based not-for-profit antiviral drug discovery company with a proven track record of advancing candidates from concept to clinical trials. Our team has successfully delivered multiple first-in-class antivirals into human studies. We offer extensive expertise in target validation, hit-to-lead, lead optimization, and early clinical strategy across various viral diseases, including HCV, RSV and flu
Current Research Focus Areas
Our core focus is the discovery and early development of novel small-molecule antivirals. Although we do not currently have assets within the scope of this call, we are actively seeking collaborations in which our established drug discovery and clinical development expertise can contribute to new antiviral modalities.
What We’re Seeking in Teaming Partners
We are looking for partners with innovative antiviral concepts that align with this call, such as novel targets, delivery technologies, or platform screening systems, and with access to relevant biological models. We aim to complement these capabilities with our expertise in medicinal chemistry, virology, and clinical development.
HMH- Center for Discovery and Innovation
Last updated:
Contact
Location
Nutley, NJ
Organization Type
- Nonprofit
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
Assets: The Metropolitan Antiviral Drug Accelerator (MAVDA) has several promising assets at various stages of preclinical development. Capabilities: The MAVDA operates pharma-style drug discovery and development platform led by directors with deep academic–industry expertise. Capabilities include virology, high-throughput screening, structural biology and computational modeling, medicinal chemistry, pharmacology, and in vivo animal models.
Current Research Focus Areas
MAVDA’s scientific focus is the discovery and development of small-molecule antiviral therapeutics suitable for outpatient settings, targeting high-threat viruses through both established and novel mechanisms of action. This scope aligns closely with the current SMART program, with relevance to viruses in the Togaviridae and Flaviviridae families. The MAVDA program has several assets starting from hit identification to near IND-ready candidates.
What We’re Seeking in Teaming Partners
We welcome partnerships that combine complementary scientific expertise and capabilities, including target biology and validation, innovative assay platforms, lead compounds or chemical series, and enabling technologies that can accelerate progression through preclinical development. In addition, we are interested in collaborations with organizations seeking a strong translational partner capable of de-risking programs and positioning them for regulatory engagement and downstream clinical advancement through effective academic–industry collaboration models.
Chung Lab: University of Louisville
Last updated:
Contact
Location
Louisville, KY
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
1. Alphavirus program: 1) Comprehensive and robust in vitro assays and mouse models for antiviral evaluation of encephalitic and arthritogenic alphaviruses, including a novel whole body imaging-based arthritogenic alphaviruses. 2) Early stage anti-CHIKV lead candidate 2. Flavivirus program: 1) Comprehensive and robust in vitro assays for flaviviruses (DENV/WNV/JEV/YFV). 2) A novel anti-Zika virus NS4B lead ( MW < 300, EC50 < 100 nM, SI50 > 1000, > 1000-fold reduction at 1 µM).
Current Research Focus Areas
Alphavirus program : 1) expanding the antiviral spectrum , 2) establishing nsP1 as a novel antiviral target. Flavivirus program : 1) lead optimization of the novel ZIKV-NS4B lead. 2) expanding the antiviral spectrum, 3) harnessing the PRO-TAC.
What We’re Seeking in Teaming Partners
1) Novel hit discovery platform(eg., DEL-Tec), 2) protein and structural biology expertise for difficult proteins including membrane proteins, 3) PRO-TAC expertise
Omphalos Lifesciences Inc
Last updated:
Contact
Location
Dallas, TX
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Flaviviridae
Expertise
- AI / ML
- Development / ADME / Safety
Relevant Technology
We are developing a mechanistic simulation platform that executes full host pathways to model viral infection, host response, and host-directed interventions. Our virtual cells combine biological rules with public and proprietary datasets, and provide quantitative insight into how interventions propagate through the system. The platform can efficiently explore combinatorial antiviral strategies at scale when multi-factor wet-lab testing becomes impractical.
Current Research Focus Areas
Our focus is host-directed antiviral design using executable whole-cell models. We build pathway-complete virtual cells to predict efficacy, toxicity boundaries, and combination behavior, replacing large experimental campaigns. We previously ported a published SARS-CoV-2 mechanistic model onto our platform and will extend this approach to dengue and Flaviviridae-relevant immune and hepatocyte contexts.
What We’re Seeking in Teaming Partners
We seek partners with dengue infection capability and small-molecule execution to generate targeted datasets for calibrating and stress-testing whole-cell models. Priority needs include: BSL-2+/3 dengue assays, THP-1 DC-like, Ramos B-cell, and HepG2 hepatocyte infection systems, host-target perturbation tools, and hepatocyte toxicity platforms. Chemistry partners able to supply and tune host-directed modulators are also desired.
Fisher Drug Discovery Resource Center of Rockefeller University
Last updated:
Contact
Location
New York, NY
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
Compound Libraries (LMW, drug-like,diverse, 600K) for High Throughput Screening;high throughput cell-based or biochemical assay development,miniaturization and automation; biophysical measurements of compound affinity, on-rates, off-rates and pharmacological concentration response curves; in vitro mechanism of action studies.
Current Research Focus Areas
Hit compound identification and hit-to-lead support.
What We’re Seeking in Teaming Partners
We are looking for partners with a specific drug target, for which they would like to interrogate with small molecule libraries; we are also interested in supporting measurement of the affinities of lead compounds in SAR studies using biophysical techniques.
Indian Institute of Science Education and Research Tirupati
Last updated:
Contact
Location
Tirupati, Andhra Pradesh
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
We have access to all relevant flavivirus isolates (Dengue virus (all four serotypes), Japanese encephalitis virus, Zika virus, West Nile virus and also togavirus (Chikungunya virus). We have cell culture models for all these isolates and also have access to animal models with collaborators. We have high-throughput infection-based screening assays in 96- and 384-well formats.
Current Research Focus Areas
Our laboratory primarily focuses on host-pathogen interactions, antiviral screening and vaccine development. We are currently employing drug repurposing strategies for Flaviviridae and Togaviridae family members. We also have colleagues in Chemistry department who have the expertise to synthesise novel derivatives of antiviral scaffolds.
What We’re Seeking in Teaming Partners
Compound libraries from either natural sources or new chemical entities with cytotoxicity data. We can also focus on the hits requiring validation after in silico screening or AI-based screening.
Drug Discovery Unit, University of Dundee
Last updated:
Contact
Location
Dundee, Scotland
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Development / ADME / Safety
Relevant Technology
The DDU is a fully integrated drug discovery research unit, bringing together assay development, screening, cell biology, medicinal chemistry, structural biology, computational chemistry and Drug Metabolism and Pharmacokinetics (DMPK) into a single integrated facility. We have 20 years of experience in delivering preclinical candidates and drugs to patients.
Current Research Focus Areas
The DDU’s mission is to successfully translate life science research into therapeutic applications to address unmet medical needs, particularly in infectious disease. We are particularly interested in the challenges of drug discovery against flaviviruses (i.e West Nile, Dengue etc) and togavirus (i.e. Chikungunya).
What We’re Seeking in Teaming Partners
The DDU is seeking partners with virology expertise to complement our drug discovery skill set. Our ideal collaborator would have the ability to perform antiviral experiments in cell-based systems and test lead molecules in in vivo efficacy/PD models. Particularly we are interested in working with collaborators who have an underexplored and exciting antiviral target they want to translate into an antiviral medicine.
READDI, Inc.
Last updated:
Contact
Location
Research Triangle, NC
Organization Type
- Nonprofit
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
READDI’s mission is to discover, coordinate, and advance antiviral medicines that can be deployed early in an outbreak. READDI is positioned to collaborate at the ‘mouth of the funnel,’ where academic discoveries, public-private collaboration, and biotechnology efforts intersect. READDI uses a portfolio capability management approach to identify promising hits and leads and provide the translational expertise required to advance them into clinical development.
Current Research Focus Areas
READDI proposes to provide centralized management for antiviral therapeutics. Rather than outfitting and operating proprietary laboratories, READDI will integrate an efficient ecosystem of innovators and service providers. This structure creates a highly flexible, capital-efficient framework capable of advancing multiple programs in parallel.
What We’re Seeking in Teaming Partners
READDI would like to collaborate with medicinal chemistry hubs, CRO‑based DMPK/toxicology platforms, virology and epidemiology expertise, biotechnology collaborators, geographically diverse regulatory and clinical networks, and domestic and global manufacturing partners.
Ginkgo Bioworks
Last updated:
Contact
Location
Boston, Massachusetts
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Development / ADME / Safety
Relevant Technology
Ginkgo Bioworks' Datapoints team offers CRO services for high-throughput, multimodal ADME-tox profiling, including DRUG-seq and cell painting readouts for perturbation response profiling analysis, as well as other essential ADME-tox endpoint assays like microsomal stability, kinetic solubility, and p450 inhibition; with the ability to onboard new assays as needed. Ginkgo has onboarded dozens of cell lines to its platform, and returns data with structured, standardized AI/ML-ready data formats.
Current Research Focus Areas
Ginkgo focuses on automated, high-throughput workflows to generate structured data sets for clients' drug development campaigns, with particular focus on ADME-tox properties of small molecule drugs. We can support Stage 1 and Stage 2 (hit-to-lead and lead optimization) proposals.
What We’re Seeking in Teaming Partners
Seeking partners with drug candidates suitable for proposals for stages 1 and 2 (and possibly Concept stage as well, to support early discovery efforts), where we can contribute data generation services.
University of Leuven
Last updated:
Contact
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
The Laboratory of Virology & Antiviral Research (www.antivirals.be) discovers and develops small-molecule antivirals. It operates an extensive integrated preclinical discovery pipeline (which led e.g. to Mosnodenvir, a highly potent pan-dengue inhibitor active in humans). We have extensive virus collections, high-throughput biosafety robotics, advanced ex-vivo and animal models, and are supported by CD3’s (www.cd3.be) large compound library and drug discovery expertise.
Current Research Focus Areas
The KU Leuven antiviral drug discovery program focuses on discovering and optimizing small-molecule direct-acting antivirals for advanced preclinical or early clinical development. It aims to identify novel druggable targets in viral replication and integrates high-throughput phenotypic screening with targeted medicinal chemistry to generate and refine lead series with broad-spectrum potential across key viral families, including Togaviridae and Flaviviridae.
What We’re Seeking in Teaming Partners
To further strengthen its potential and accelerate progression toward IND qualification and beyond, the KU Leuven team is seeking partnerships and expertise in structural biology of viral proteins (X-ray crystallography, cryo-EM) to enable high-resolution target characterization and structure-guided design, GLP/GMP production and CMC to ensure scalable and regulatory-compliant manufacturing of lead compounds, comprehensive toxicity and safety assessment, including both in vitro and in vivo models, to support preclinical (GLP Tox) and regulatory filings.
ARIScience
Last updated:
Contact
Location
Wayland, MA
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
5 DENV antiviral hits with potential for applicability towards WNV, JEV, ZIKA
Current Research Focus Areas
We plan to perform SAR, PK, toxicity/off-target effect and efficacy studies focused around our 5 actionable DENV antiviral hits
What We’re Seeking in Teaming Partners
We are looking for collaborators to perform med-chem and SAR focused around our 5 actionable DENV antiviral hits followed by toxicity, off-target effect, PK and efficacy studies
X-Chem, Inc.
Last updated:
Contact
Location
Waltham, MA
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
X-Chem is redefining small molecule drug discovery through a powerful combination of computational sciences, discovery chemistry and data-driven insight. As the pioneer and global leader in DNA-encoded library (DEL) technology, X-Chem provides the foundation for smarter hit identification, accelerated lead progression and greater confidence in early discovery.
Current Research Focus Areas
X-Chem has recently developed Chemomics, an integrated approach to creating, analyzing, and applying omics-scale chemistry data, ensuring every data point drives discovery forward. We transform full DEL screen data into structure-activity roadmaps, mechanism-of-action insight and synthesis-ready design recommendations.
What We’re Seeking in Teaming Partners
We are looking for partners who need unique and timely chemistry solutions their viral drug discovery programs from a provider incentivized by success rather than hourly rates.
Institute for Antiviral Research, Utah State University
Last updated:
Contact
Location
Logan, UT
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
The Institute for Antiviral Research (IAR) at Utah State University has been conducting antiviral studies in relevant cell culture and animal models of flaviviruses and togaviruses for over 20 years. Previous work has been focused on providing preclinical data, including antiviral efficacy and pharmacokinetic studies, to assist compound suppliers in preparing an IND for further development. We have accredited BSL/ABSL-2 and -3 and select agent facilities.
Current Research Focus Areas
The focus of our work is modeling the replication and disease of various flaviviruses and togaviruses in relevant small animal models for the purpose of testing the efficacy of various antivirals and vaccines. We have established rodent models of WNV, YFV, ZIKV, CHIKV, MAYV, VEEV, EEEV, and WEEV and have demonstrated the efficacy of various antivirals and vaccines in these models. These data have been used to move countermeasures forward towards clinical development.
What We’re Seeking in Teaming Partners
We seek partners in medicinal chemistry that would like to test the efficacy of their compounds in relevant, established models of flavivirus and alphavirus disease to generate preclinical data for the support of IND submission. Additional partners may include laboratories with formulation capabilities to improve the pharmacokinetic profile of compounds for enhanced delivery of antiviral compounds that show promise in antiviral experiments.
National University of Singapore
Last updated:
Contact
Location
Singapore, Singapore
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
The NUSMed BSL-3 Core Facility is a unit in National University of Singapore conversant with infectious disease preclinical testing and mechanism discovery. Our unit is equipped with resources to conduct preclinical screenings of antivirals of different nature, including small molecules, peptide antivirals and natural products. We are a facility with both ABSL-2 and ABSL3 capabilities for in vitro and in vivo preclinical testing, providing evaluation up to small animal validation.
Current Research Focus Areas
The NUSMed BSL-3 Core Facility specialty includes antiviral drug screening and lead compound optimization, as well as molecular mechanism discovery of the drug interaction with the virus in vitro and in vivo. We have experience working with viruses from both Togaviridae and Flaviviridae families, at both BSL-2 and BSL-3 containment levels. Our unit has completed drug screening and preclinical follow up in vitro and in vivo of selected antiviral compounds and its lead derivatives.
What We’re Seeking in Teaming Partners
We seek teaming partners who have antiviral hits or lead compounds or varying nature (small molecules, peptides etc) who would like to follow up with preclinical in vitro and in vivo (small animal) validation of the compound activity on selected viruses from the Togaviridae (e.g. Chikungunya) and Flaviviridae (e.g. Dengue, West Nile, Zika) family. The validation may include antiviral activity assays and mechanisms discovery in BSL-2 and -3 settings.
Texas Biomedical Research Institute
Last updated:
Contact
Location
San Antonio, TX
Organization Type
- Nonprofit
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
Texas Biomed offers an integrated antiviral platform combining high-throughput in vitro screening (plaque, RT-qPCR, reporter assays) with validated small animal and nonhuman primate models in ABSL-3/4. As a Select Agent, we support high-consequence pathogens under strict biosafety standards. Our GLP-aligned assays and FDA Animal Rule experience enable rapid, regulatory grade antiviral evaluation from screening through efficacy, PK/PD and translational decision making. We welcome partnerships.
Current Research Focus Areas
Primary focus is antiviral evaluation using integrated in vitro assays and validated small animal and NHP models in BSL-3/4. We support work on filoviruses, arenaviruses, alphaviruses, flaviviruses, coronaviruses, and other high-consequence pathogens. Our platform is mature and operational, with GLP-aligned assays and FDA Animal Rule experience, enabling immediate support for candidate screening, efficacy, PK/PD studies and translational decision making.
What We’re Seeking in Teaming Partners
Texas Biomed offers fully established BSL-2/3/4 facilities, validated animal models, and GLP-aligned assay capabilities. With appropriate partners and funding, we can develop, optimize, and validate disease models and analytical assays tailored to specific antiviral candidates. Our team provides end-to-end support from model development through efficacy testing and regulatory-ready data generation.
Emerging Pathogens Institute - University of Florida
Last updated:
Contact
Location
Gainesville, Florida
Organization Type
- Academia
Virus Family of Interest
- Flaviviridae
Expertise
- AI / ML
- Virology & Infection Models
Relevant Technology
EPI is an interdisciplinary research institute at the University of Florida, Gainesville, focused on the emergence and control of human, animal and plant pathogens that impact Florida, the nation and the globe. Over the past five years, we have developed a robust research program to apply Artificial Intelligence/machine learning (AI/ML) algorithms to investigate and forecast the evolution of viral pathogens (primarily focusing on coronaviruses, flaviviruses, and immunodeficiency viruses).
Current Research Focus Areas
AI/ML models development to predict the evolution of human and animal viruses; application of AI/ML to investigate and forecast drug resistance at the genomic level; development of models that can predict the spread of infectious diseases and use of causal AI models to investigate the impact and implementation of prevention and treatment strategies.
What We’re Seeking in Teaming Partners
EPI has access to the University of Florida HiPerGator, the largest high performance computer cluster among US academic institutions. We have strong expertise in ML/AI models development and implementation, and aim to partner with academic and private institutions with a track record on novel antivirals.
Evotec
Last updated:
Contact
Location
Hamburg, Germany
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
Evotec is a global life science company providing end-to-end drug discovery and development services, from target ID to clinical and commercial manufacturing. It combines cutting-edge science, AI-driven innovation and advanced technologies to accelerate progress from concept to cure. With deep virology expertise, integrated discovery capabilities, translational ADME/PK, and robust mechanistic and in-vivo validation, Evotec delivers high-quality data to provide fast, precise R&D decisions.
Current Research Focus Areas
Evotec accelerates first-in-class antiviral programs using disruptive small molecule modalities, including RNA-targeting and targeted protein degradation. Backed by deep virology expertise across respiratory, chronic, vector-borne, and emerging viruses, Evotec balances direct acting and host targeting strategies to deliver broad, resistance robust antiviral solutions.
What We’re Seeking in Teaming Partners
Evotec supports partners to accelerate next-generation broad-spectrum antivirals through two collaboration models:
• Collaborations focused on identifying novel viral targets and applying innovative approaches to develop first-in-class, resistance-resilient therapeutics.
• Collaboration with groups working on validated viral targets or promising small molecules seeking access to Evotec’s discovery capabilities and extensive expertise.
• Collaborations focused on identifying novel viral targets and applying innovative approaches to develop first-in-class, resistance-resilient therapeutics.
• Collaboration with groups working on validated viral targets or promising small molecules seeking access to Evotec’s discovery capabilities and extensive expertise.
Virology Research Services Ltd
Last updated:
Contact
Location
Sittingbourne, Kent, United Kingdom
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
We are a UK-based contract research organisation specialising in in-vitro antiviral evaluation. Our team has deep expertise in alphaviruses and flaviviruses, including dengue, Zika and Ross River virus. We deliver studies ranging from routine potency screening to customised programmes, such as resistance selection and mechanism-of-action analysis, with flexible assay development and reliable turnaround.
Current Research Focus Areas
Primary focus is in-vitro antiviral testing and bespoke assay development. We operate as an established service provider with CL2 and CL3 capabilities, validated platforms, and experienced staff, running validated workflows and customised projects for external R&D programmes.
What We’re Seeking in Teaming Partners
While we are not intending to submit an application or participate as an equal consortium partner, we are keen to support applicants who require high-quality in-vitro antiviral testing, assay development or mechanistic studies. We welcome engagement with groups that have candidate compounds, platforms or concepts needing experimental evaluation in validated alphavirus and flavivirus systems.
Gavande Lab - Wayne State University
Last updated:
Contact
Location
Detroit, MI
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Development / ADME / Safety
Relevant Technology
Dr. Gavande trained with the distinguished medicinal chemist Prof. Arun K. Ghosh, whose laboratory developed Darunavir (Prezista®), an FDA-approved first-line therapy for HIV/AIDS. Dr. Gavande interdisciplinary research focuses on the preclinical development of novel antiviral, antibacterial and anticancer therapeutics. His team integrates medicinal chemistry, structure-based drug design, molecular modeling, and AI-driven approaches to accelerate drug discovery.
Current Research Focus Areas
The Gavande laboratory is actively engaged in antiviral drug discovery, including the development of inhibitors targeting viral proteases and host-dependent pathways that regulate viral replication. These studies are designed to address emerging viral threats and drug-resistant viral infections by identifying novel molecular scaffolds and mechanisms of action.
What We’re Seeking in Teaming Partners
I am seeking collaborators who can perform comprehensive biological analyses, including enzymatic assays, antiviral activity evaluation, and in vivo studies. The goal is to advance the preclinical evaluation of candidate compounds targeting either flaviviruses or Togaviruses. Expertise in virology, viral replication assays, and animal models of viral infection would be highly valuable for this collaborative effort.
Advanced Virology Inc.
Last updated:
Contact
Location
Athens, OH
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
We use cutting-edge plasmid-based reverse genetics systems to manufacture very high-fidelity virus stocks and derivative products (e.g., reporter viruses) for use in antiviral R&D. Our de novo generated virus stocks are created directly from sequence files, are never passaged or cell adapted, and maintain genetic and phenotypic fidelity between production batches. We have extensive experience in generating large scale Togavirus and Flavivirus stocks for downstream testing.
Current Research Focus Areas
Our primary focus area is in the generation of very high fidelity virus stocks and derivative products such as reporter viruses. We focus exclusively on development and optimization of virus production methods. We currently have the capability to produce liter scale batches of both Togaviruses and Flaviviruses.
What We’re Seeking in Teaming Partners
We are looking to partner with any organization that needs virus stocks for use in their R&D work. We can onboard existing and novel virus strains as well as serve as a virus repository for the production and distribution of large scale virus stocks.
Yang Lab: Stanford University
Last updated:
Contact
Location
Stanford, CA
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
We have experience developing DAAs that have a targeted protein degradation mechanism. We are interested in collaborating with other groups to advance our existing leads including in vivo validation and optimization. We are also interested in collaborating to apply targeted protein degradation to other Flavivirus and Togavirus targets.
Current Research Focus Areas
We have validated small molecule degraders of the dengue virus envelope and capsid proteins.
What We’re Seeking in Teaming Partners
We experience developing and characterizing direct-acting antivirals that act by a targeted protein degradation mechanism. We have lead molecules for dengue virus that are well-characterized in cell culture that need to be optimized for antiviral potency and ADME/PK to advance and are seeking partners with synthetic chemistry capacity and experience in humanized mouse models for evaluating antivirals. We are also interested in collaborating with other groups who have validated ligands of Flavivirus and/or Togavirus proteins and who are interested in turnings these into degraders.
NEIDL: Boston University Medical Campus
Last updated:
Contact
Location
Boston, Massachusetts
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
We have unique capabilities through the National Emerging Infectious Diseases Laboratories to characterize the efficacy of antiviral compounds at BSL2, BSL3, and BSL4 in both cell culture and animal models (from mouse to non-human primates). To support this, we also develop recombinant orthoflavivirus and alphavirus reporter clones and replicons, and iPSC-derived human organoids. We also have an ACL2/3 insectary with active mosquito colonies to perform mosquito-to-mouse transmission studies.
Current Research Focus Areas
We are a cadre of RNA virologists and cell biologists with established research programs on 1) Orthoflavivirus and alphavirus molecular biology and pathogenesis; 2) Low-mid throughput antiviral compound screening and mechanism of inhibition studies of antivirals; 3) Development of iPSC-derived organoids covering a wide range of human tissues to characterize viral infections; and 4) Animal model development for virology, from rodent models and humanized mice to mosquitoes and non-human primates.
What We’re Seeking in Teaming Partners
We are uniquely positioned to support partners with molecule-discovery capabilities in evaluating the antiviral properties of their compounds through a comprehensive preclinical pipeline. We are looking to collaborate with partners who: 1) Have antiviral discovery methods and look to perform initial in vitro efficacy studies; 2) Have candidate antivirals for organoid and in vivo efficacy studies; and 3) Have established candidates for advanced in vivo validation studies.
CreaGen Inc.
Last updated:
Contact
Location
Woburn, MA
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
Relevant Technology
CreaGen has strong expertise in antiviral drug design and medicinal chemistry, supporting programs from hit identification through lead optimization. We have designed and synthesized diverse small-molecule antivirals using complex, multi-step chemistry and rapid SAR development. CreaGen collaborates with leading academic and industry partners, including The Scripps Research Institute, and currently holds an active NIAID contract. We work with NIAID’s antiviral and pandemic preparedness program.
Current Research Focus Areas
CreaGen’s primary focus is small-molecule drug discovery and medicinal chemistry, with strengths in rational drug design, synthetic chemistry, and structure–activity relationship development. Our work spans hit identification through lead optimization, including analog design and scalable synthesis of complex molecules. CreaGen currently operates at the preclinical discovery and lead optimization stage, supporting academic, biotechnology, pharmaceutical, and federal research programs worldwide.
What We’re Seeking in Teaming Partners
We are seeking a biology collaborator who has identified chemical matter (e.g., via screening) and needs chemists to design and synthesize improved analogs. This collaborator should also be able to rapidly test the newly synthesized analogs and report data.
Pop Test Oncology LLC Aka Palisades Therapeutics
Last updated:
Contact
Location
Cliffside Park, NJ
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Virology & Infection Models
Relevant Technology
PT therapeutics are host-directed small molecules that modulate glucocorticoid and androgen receptor signaling to reduce viral entry factors, inhibit replication, and increase resistance barriers across multiple viral families. Unlike many teams that primarily provide discovery or CRO services, Pop Test Oncology develops viable therapeutic product candidates, with a platform that includes clinical-stage PT150 and next-generation antiviral candidates PT155 and PT157 for broad-spectrum infectious disease and biodefense applications. PT155 is being developed as an oral antiviral for flaviviruses including dengue, Japanese encephalitis, West Nile, and Powassan, while PT150 provides a de-risked clinical foundation with completed Phase 1/2 experience, prior COVID-19 IND activity, and cGMP manufacturing history.
Current Research Focus Areas
Company develops PT-series host-directed antivirals and related small-molecule therapeutics targeting infectious disease, biodefense, oncology, CNS disorders, and other high-unmet-need indications. Current infectious disease focus is antiviral development led by PT150, expanded by preclinical PT155 for stockpile-ready broad-spectrum flavivirus defense, and supported by PT157 as an additional follow-on preclinical antiviral candidate in the same platform family. PT155 has demonstrated broad live-virus activity in NIAID-supported testing and is being advanced toward hit-to-lead optimization and IND-directed development.
Additional Platform Strength : Company also has TPR1, a rifamycin-derived antibacterial with recently granted European composition-of-matter protection, existing U.S. protection, and related derivatives PT159 and PT160. TPR1 is currently in Phase 2 USDA-funded replicated grove trials for citrus greening, underscoring broad anti-infective platform depth and extensive intellectual property coverage.
Additional Platform Strength : Company also has TPR1, a rifamycin-derived antibacterial with recently granted European composition-of-matter protection, existing U.S. protection, and related derivatives PT159 and PT160. TPR1 is currently in Phase 2 USDA-funded replicated grove trials for citrus greening, underscoring broad anti-infective platform depth and extensive intellectual property coverage.
What We’re Seeking in Teaming Partners
Pop Test Oncology seeks strategic partners with expertise in antiviral drug development, medicinal chemistry and SAR optimization, in vivo pharmacology, ADME/PK, GLP toxicology, regulatory strategy, and commercial manufacturing scale-up for oral small molecules. The company also seeks collaborators with biodefense and government contracting relationships, including organizations aligned with BARDA, DTRA, and DARPA. Licensing, co-development, and strategic collaboration discussions are welcome.
Collaborations Pharmaceuticals, Inc.
Last updated:
Contact
Location
Raleigh, North Carolina
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
We developed a number of software (AI) tools to design and identify molecules with desired bioactivity and ideal ADME/Tox properties. These include our software for generative molecule design (MegaSyn), custom machine learning model building (Assay Central), ADME/Tox models (MegaTox) and large collections of human and pathogen targets (MegaPredict). We have applied several of these tools to our antiviral and other projects including Yellow fever virus.
Current Research Focus Areas
Our primary focus is on the continued development of these tools and applying them to our own and other companies projects to further validate them. Our most advanced antiviral project is for Ebola Virus and this molecule was identified using machine learning and is currently at the stage of testing in non-human primates.
What We’re Seeking in Teaming Partners
Antiviral testing in vitro and animal models
Integrated Biotherapeutics LLC DBA IBT Bioservices
Last updated:
Contact
Location
Rockville, MD
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
IBT is a specialized Infectious Disease CRO dedicated to advancing global health by providing expert R&D services for development of vaccines and therapeutics. We have validated, ready-to-go Virus Neutralization Assays, Immunoassays, Animal Efficacy & Safety Models to support preclinical development. With a robust QMS, we can also support end-point assays for clinical sample testing. We also produce custom quality reagents like viral glycoproteins, VLPs, pseudovirus, antibodies & more.
Current Research Focus Areas
As a full-service nonclinical CRO, we are experts in Flaviviruses and Togaviridae. Operating BSL-2 labs, we provide end-to-end functional screening via viral neutralization and multiplex assays. We deliver GCLP-compliant clinical sample testing backed by expert assay qualification and the production of high-quality reagents, including VLPs and antibodies. Our OLAW-approved rodent, hamster, and guinea pig models support efficacy, PK, biodistribution, and safety evaluations.
What We’re Seeking in Teaming Partners
To strengthen our offerings, we seek partners with complementary specialized expertise in high-containment pathogens, specifically regarding BSL-3/4 capabilities and large animal model access. We value organizations with advanced platform technologies for rapid vaccine discovery and AI-driven lead optimization. Ideal collaborators offer unique clinical infrastructure or niche regulatory experience to bridge the gap between nonclinical efficacy and Phase I readiness, ensuring a seamless translational path.
Living In Silico Inc.
Last updated:
Contact
Location
Brampton, Ontario, Canada
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Flaviviridae
Expertise
- Discovery & Design
- AI / ML
Relevant Technology
We aim to utilize an AI/Machine learning model to predict compounds for their anti-Flaviviridae activity. In addition, we will deploy a toxicology prediction model to further identify candidates that are screened.
Current Research Focus Areas
Ideation stage. The primary focus area of our work is drug discovery and design using AI.
What We’re Seeking in Teaming Partners
AI/Machine learning expertise and domain knowledge
University of Washington
Last updated:
Contact
Location
Seattle, WA
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
The Polyak Laboratory has decades of experience on in vitro antiviral testing of drugs, both synthetic and natural products, against diverse RNA virus families including flaviviruses (PMID: 29895962; 20231449) and togaviruses (PMID: 40379030), the focus of the SMART funding call. Available assays include cytopathic effect (CPE)-based for general antiviral screening plus orthogonal assays that assess viral RNA, protein, and infectious virus production. MOA studies are also available.
Current Research Focus Areas
We focus solely on in vitro characterization of drugs that directly target viral proteins/enzymes (DAA) as well as the potential for host-directed antiviral (HTA) compounds. We collaborate extensively with virologists who possess animal model systems that evaluate the in vivo potential of lead molecules. Independent of the SMART funding call, we are part of a larger consortium of academic and industrial teams called VORTEC that focuses on drug combinations for emerging and re-emerging viruses.
What We’re Seeking in Teaming Partners
Please see the prior section for our in vitro antiviral testing capabilities. We are highly collaborative and seek to work with teams who have novel compounds and animal models as well as teams who consider drug behavior (PK/PD) and viral kinetic models.
BIOQUAL, Inc.
Last updated:
Contact
Location
Rockville, Maryland
Organization Type
- Service Provider (CRO, CDMO)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
- Development / ADME / Safety
Relevant Technology
Established in 1981, BIOQUAL’s technologies encompass comprehensive preclinical animal models for infectious and non-infectious diseases in rodents, small non-rodent animals, and NHPs; advanced in vitro and 3D cell culture assays; BSL-2/3 biocontainment; non-invasive whole-body imaging; GLP-compliant IND-enabling studies; and extensive government and industry contract experience.
Current Research Focus Areas
Our primary focus is supporting the development and evaluation of medical countermeasures for emerging and high-consequence infectious diseases, including vaccines, monoclonal antibodies, antivirals, and other therapeutics. We provide in vivo efficacy models, advanced in vitro and 3D culture systems, and immunological and molecular assays across preclinical through IND-enabling studies to generate high-quality, decision-enabling data.
What We’re Seeking in Teaming Partners
BIOQUAL seeks partnerships with organizations pursuing cutting-edge antiviral solutions that require preclinical testing and infectious disease challenge models. We support antivirals, vaccines, and therapeutics through exploratory in vivo infectious disease studies in ABSL-2 and ABSL-3 settings, complemented by advanced in vitro and 3D culture systems, as well as immunological and molecular assays, to generate decision-enabling data.
Biomolecular Science, LLC
Last updated:
Contact
Location
Huntsville, Alabama
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
Expertise
- Discovery & Design
- Development / ADME / Safety
Relevant Technology
Our antiviral development program is focused on small‑molecule antivirals against alphaviruses, with a central focus on the nsP2 cysteine protease as a validated therapeutic target. Candidate molecules active against multiple alphaviruses, including VEEV, WEEV, MAYV, and CHIKV, are now in preclinical evaluation. We have recently completed experiments that show one of our broad-spectrum inhibitors has therapeutic efficacy in a lethal mouse model of VEEV infection.
Current Research Focus Areas
Current Focus: Alphavirus Drug Discovery - Lead Optimization and ADME, PK. Current stage of development: Late-stage lead optimization and prospective NHP studies.
What We’re Seeking in Teaming Partners
a) Non-human primate studies capability
b) Pre-IND and IND enabling safety pharmacology and toxicology studies and review capability
c) Large-scale API synthesis, characterization, and formulation capability
d) cGMP compliance and quality systems capability
b) Pre-IND and IND enabling safety pharmacology and toxicology studies and review capability
c) Large-scale API synthesis, characterization, and formulation capability
d) cGMP compliance and quality systems capability
Xie Lab: The University of Texas Medical Branch
Last updated:
Contact
Location
Galveston, TX
Organization Type
- Academia
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Virology & Infection Models
Relevant Technology
1) Cell-based reporter virus assays: diverse, validated reporter virus–derived antiviral screening across Flavivirus and Togaviridae, with counterscreening in additional viral families at BSL-2 to -4 2) Compound libraries: >1,000,000 high‑quality, chemically diverse small molecules for screening 3) Comprehensive biochemical, biophysical, and virological assays for mechanistic validation 4) Antiviral efficacy testing in rodents for flaviviruses 5) NHP in vivo efficacy
Current Research Focus Areas
Focus areas: Cell-based and target-based high-throughput screening; in vivo antiviral efficacy testing; target deconvolution and mechanistic studies. Development stages: our prior work in UNAPP (AVIDD Center) has generated hits, leads, and drug candidates for flaviviruses across multiple stages of development. Several candidates have demonstrated efficacy in rodents with favorable safety profiles. Ongoing work includes novel antiviral strategies to identify new hits.
What We’re Seeking in Teaming Partners
1) Medicinal chemists for drug discovery design & lead optimization; 2) AI and ML experts for drug design and profiling; 3) Development and safety, drug formulation/manufacturing/regulatory
Brimrose Technology Corporation
Last updated:
Contact
Location
Sparks, Maryland
Organization Type
- Startup (Biotech, AI)
Virus Family of Interest
- Togaviridae
- Flaviviridae
Expertise
- Discovery & Design
- Development / ADME / Safety5
Relevant Technology
We have research experience on drug discovery and development against viruses including HIV, EV71, and SARS-COV2. We previously developed software solutions for search of affinity molecules against pathogenic viruses of pandemic potential.
Current Research Focus Areas
Focus areas include protein affinity analysis in silico and with biochemical and biophysical approaches.
What We’re Seeking in Teaming Partners
AI and clinical resources and providers.
Disclaimer
All information provided within is preliminary and subject to change. Specific criteria and requirements are currently being refined. In the event of any inconsistency, the official SMART Antiviral Prize Terms & Conditions supersede this information.
Program Overview and Rationale
BARDA currently supports multiple efforts to develop therapeutics against several biological threats. One consistent limitation in current medical countermeasure (MCM) preparedness efforts is the lack of successful processes and approaches available to improve the early-stage pipeline for small molecule therapeutics. This aspect of MCM preparedness is particularly challenging given that traditional approaches have not worked well for these types of therapeutics.
Therefore, the goal of this effort is to identify new technical approaches that can address this critical gap in preparedness. Under this effort, approaches are sought that can produce new, safe, and effective small molecules active against a broad range of viruses within one or more viral families. The SMART Antiviral Prize will be composed of four stages, with success in the final stage culminating in an IND-ready candidate prepared to progress to Phase I clinical trials.
What are the main goals of the SMART Antiviral Prize?
We seek to award new innovators and new breakthrough approaches, while encouraging participation from both traditional and non-traditional innovators in order to expand the pool of creative and feasible solutions. The goals of the SMART Antiviral Prize are to address gaps in the current pipeline of therapeutics that can advance into the clinic, as well as identify promising approaches to small molecule development that could be rapidly applied to future threats.
Why is BARDA using a prize competition structure to support innovative small molecule antiviral development?
As outlined in the Program Overview and Rationale section, BARDA recognizes the importance of broad-spectrum small molecule antiviral approaches that strengthen the therapeutic pipeline and address critical preparedness gaps. The prize competition model encourages participation from both traditional and non-traditional innovators, expanding the pool of creative and feasible solutions.
Scientific Scope & Eligibility
What specific types of antivirals are allowed?
The SMART Antiviral Prize is limited to traditional small molecule drugs—organic compounds with a molecular weight at or below 900 Daltons that can be chemically synthesized or isolated from natural sources (e.g., plants, animals, minerals). The following are not permitted: biologics (including peptide-based products and antibody–drug conjugates) and nucleic acid-based drugs. Nucleotide and nucleoside analogs are allowed and considered traditional small molecule drugs for the purposes of this prize competition.
What mechanisms of action are allowed?
Proposed antivirals must directly target highly conserved viral factors (direct-acting antivirals) or proviral host factors required for viral entry, replication, or persistence (indirect-acting antivirals). Products without measurable antiviral activity will not be considered. “Measurable antiviral activity” refers to demonstrated inhibition of viral replication or reduction in viral load in relevant in vitro and in vivo models. Products that act solely through immunomodulatory or symptomatic mechanisms without directly impacting the viral lifecycle will not be considered.
Are clinical-stage compounds eligible?
No. Compounds that have been tested in humans at any stage of clinical development (Phase I–IV) are not eligible for this prize. This includes FDA-approved drugs, repurposed clinical-stage compounds, and reformulations of approved or previously clinical-stage drugs. Chemical derivatives of such would be eligible if they are New Chemical Entities, with substantive molecular modification beyond reformulation, and applicants must demonstrate appropriate freedom to operate (FTO) when leveraging proprietary compounds as starting points.
Developers with more advanced products are encouraged to review open solicitations to identify potential alternative opportunities to apply for BARDA funding.
What viral families are within scope?
The SMART Antiviral Prize focuses on identifying antivirals with broad-spectrum activity against the Togaviridae and/or Flaviviridae families.
Both viral families are prioritized by the Public Health Emergency Medical Countermeasures Enterprise (PHEMCE) due to their potential to emerge as novel, high-consequence pathogens capable of causing a U.S. public health emergency. In addition, these viral families were selected based on alignment with BARDA’s published Emerging Infectious Diseases (EID) strategy (Johnson et al. 2025) and prioritization frameworks (White et al. 2025; White et al. 2025). Specifically, BARDA’s prioritization framework emphasizes factors such as public health impact, outbreak size and frequency, technical feasibility of development, and readiness of countermeasure approval pathways.
Is it expected that high-containment viruses must be tested?
No. Preclinical results are expected to be confirmed using live, non-attenuated viruses or strains, which may include BSL-3 and/or select agents. BSL-4 viruses or strains will not be required due to facility access limitations.
What is the primary indication for use?
The antiviral candidate should be suitable for treatment of acute, laboratory-confirmed infection caused by viruses within the Flaviviridae and/or Togaviridae families.
Is oral administration absolutely required? Can an entrant focus on developing an antiviral for other routes of administration?
The SMART Antiviral Prize is intended to advance orally deliverable antiviral candidates toward the clinic. At the Concept Stage, entrants are not required to provide oral exposure or dosing data. However, the development plan should describe how the candidate could be advanced toward oral administration, including any work needed to improve oral bioavailability and related properties. Feasibility data supporting oral exposure and dosing are expected in later stages of the prize. Refer to the Desired Products Attributes for additional information.
Can an entrant propose a combination therapy for the SMART Antiviral Prize?
No, combination therapy is out of scope and will be ineligible.
If an entrant has a molecule with promising preclinical data, but it has not yet been tested against togaviruses or flaviviruses, are they still eligible to apply for the Concept Stage?
Yes. Applicants may apply to the Concept Stage even if their molecule has not yet been specifically tested against togaviruses or flaviviruses, provided they otherwise meet the applicable eligibility requirements for the prize. Applicants should review the eligibility criteria carefully and ensure their submission aligns with all Concept Stage requirements.
Eligibility and Participation Rules
Who is eligible to apply?
The Primary Entrant must be a U.S.-based Concern at the time the Notice of Intent is submitted and remain a U.S.-based Concern through the issuance of prize awards.
“U.S.-based Concern” means: (i) incorporated in the United States (including a U.S.-incorporated subsidiary even if the parent is foreign based), or (ii) unincorporated with its principal place of business in the United States. Principal place of business is defined as the primary location where management directs, controls, or coordinates the entity’s activities.
Note: Components of the work may be performed by a team consisting of subcontractors, consultants, CROs, or partner organizations, however, the Primary Entrant must retain responsibility for substantively leading the technical effort.
See the Terms & Conditions here.
Does an applicant need to be at a certain stage of development to be eligible for the Concept Stage?
Eligibility requirements can be found in the section above. Concept Stage application details can be found on the Prize Details page. There is no minimum Technology Readiness Level requirement.
Is participation in a prior stage(s) required to be eligible for entry into a subsequent stage?
No. Stages beyond the Concept Stage will be open to previous entrants, prior prize winners, and new entrants.
If an entrant applies to—but does not receive the prize for—a given stage, are they eligible to apply for a subsequent stage?
Yes. An entrant may enter or re-enter the prize competition at any subsequent stage, provided they meet the eligibility criteria for that stage. For Stages 1–3, entrants must also submit and receive approval of a Declaration of Intent to be eligible for prize awards.
Can an entrant submit more than one submission for the same stage?
Yes. However, an entrant may only be selected as the prime awardee for one award per stage.
What are the submission review criteria?
Submission review criteria will be published at the opening of each stage. Submission review criteria for the Concept Stage can be found here.
What are the reporting requirements?
Reporting requirements specific to each prize stage will be released at the opening of each stage. For the Concept Stage, winners will have quarterly meetings with VITAL, in which the winner will provide updates on the following topics as they pertain to the proposal: progress on development toward the goals of the proposal, technical achievements and milestones, technical troubleshooting, risks and mitigation planning, intellectual property, regulatory strategy and planning, changes in personnel and cybersecurity updates. Quarterly meetings will follow a template, which will be provided to the winners.
Do I need a SAM.gov registration?
No.
Are federal government (USG) employees and USG support contractors eligible to participate in submissions?
No. USG employees and support contractors are not eligible to be part of a submission in any capacity – whether as primary submitters, team members, collaborators, advisors, or any other role, and whether paid or unpaid.
Are foreign subcontracts/collaborators permitted?
The primary applicant/award recipient must be a U.S. based concern. Foreign subcontractors and teaming partners may be included, provided they comply with the eligibility requirements. See the Terms & Conditions here.
Does an applicant already need to be a U.S. based entity at the time of submitting the concept stage application?
Yes. The primary applicant/award recipient must be a U.S. based concern at the time of concept stage application.
May a primary entrant submit more than one application for the same stage, and can more than one award be made to the same primary entrant?
Yes. A primary entrant may submit more than one application for the same stage, and each application will be evaluated independently. For entrants other than academic institutions, however, only one award may be made per stage to the same primary entrant. If multiple submissions from the same non-academic primary entrant are among the highest-evaluated submissions, only the highest-evaluated submission will be selected for award. Separate teams, laboratories, business units, or investigators within the same non-academic organization will not be treated as separate primary entrants for purposes of award selection.
A limited exception applies to academic institutions. Multiple applications from different Principal Investigators (PIs) at the same academic institution may be considered for, and may receive, award in the same stage, even though the institution is the legal entity that would receive and administer the funds, provided that each application represents a distinct and independent effort. Each such application must reflect a clearly different scientific approach, separate team, and distinct supporting resources, and must not overlap in personnel, scope, or underlying solution. This exception recognizes that, in academia, the institution is often the legal recipient of funds, while the scientific work may be independently developed and led by different PIs or research groups.
The primary entrant identified in the application may not be changed after submission. Note the primary entrant must also be a legal entity (such as a non-profit, for-profit, or academic institution). See the Concept Stage Evaluation Process & Criteria here.
Is the SMART Antiviral Prize funding intended to cover all project costs, or only direct costs?
The prize is a fixed award provided to competitors who are selected as winners under the competition rules. Unlike a grant or contract, a prize is not a cost-reimbursement mechanism and is not based on direct, indirect, proposed, or incurred costs. It is expected winners will use the prize funds to support further development and participation in subsequent stages of the competition, subject to the terms and conditions.
Concept Paper Submission Template
Preview the Concept Stage proposal template ahead of submitting an application. This file is for reference only and is not submittable. To submit your proposal, click “Apply Now” at the top of the page.
Evaluation Process & Criteria
The SMART Antiviral Prize uses a two-stage review process. This document outlines the Evaluation Process & Evaluation Criteria for the Concept Stage.
Desired Product Attributes
This document outlines Desired Product Attributes that applicants may find useful when shaping antiviral concepts for submission to the SMART Antiviral Prize.
Target Compound Profile
The SMART Antiviral Prize has outlined the set of target criteria for potential applicants to reach by the end of Stage 1. See this document for more information.
Info Session Recording
Watch the recording to get an overview of the prize vision, scientific scope, and key details for potential applicants. For a deeper dive and the opportunity to ask questions live, we encourage you to join one of our upcoming Info Sessions.
Launch Event Presentation
Did you miss the SMART Antiviral Launch Event? No worries – explore the presentation shared at the SMART Antiviral Prize launch, covering the challenge vision, priorities, and scientific scope.
Disclaimer
All information provided within is preliminary and subject to change. Specific criteria and requirements are currently being refined. In the event of any inconsistency, the official SMART Antiviral Prize Terms & Conditions supersede this information.
